Funding for the development of this content was provided by Pharmacyclics LLC and Janssen Biotech, Inc. The content was created by ONS, Edythe M. Greenberg, PhD, FNP-BC, CEN, CDE, and Ashley Bryant, PhD, RN-BC, OCN®, independently of Pharmacyclics LLC and Janssen Biotech, Inc.
Ibrutinib is a first-in-class Bruton’s tyrosine kinase inhibitor that is approved by the U.S. Food and Drug Administration to treat chronic lymphocytic leukemia (CLL)/small lymphocytic lymphoma (SLL). The drug’s approval was based on results from the multicenter, open-label, phase III RESONATE trial.
In a poster presented on Thursday, May 17, 2018, at the ONS 43rd Annual Congress, researchers reported on long-term follow-up from this study and discussed the critical role oncology nurses play in managing treatment-related adverse events (AEs).
The RESONATE trial enrolled 391 patients from 67 sites in the United States, Australia, and seven European countries between June 2012 and April 2013. Patients had a confirmed CLL/SLL diagnosis, received one or more prior lines of therapy, and had an Eastern Cooperative Oncology Group performance status score of 0–1.
Patients were randomized 1:1 to receive either:
- Oral ibrutinib 420 mg once daily until disease progression or unacceptable toxicity (n = 195)
- A 300 mg initial dose of IV ofatumumab followed by 2,000 mg for 11 doses over 24 weeks (n = 196)
Patients received a median of three prior therapies (range = 1–12 therapies) in the ibrutinib cohort and a median of two prior therapies (range = 1–13 therapies) in the ofatumumab cohort. The median patient age was 67 years in both cohorts (range = 30–86 years and 37–88 years, respectively). In the ibrutinib cohort, 63 patients (32%) had del17p mutation and 63 (32%) had del11q mutation. In the ofatumumab cohort, 59 (30%) and 64 (33%) patients, respectively, had the mutations.
In the ofatumumab cohort, 144 patients were eligible to crossover to the ibrutinib cohort and 133 did so.
At a median follow-up of 44 months (range = 0.07-53.0 months), the progression-free survival (PFS; primary endpoint) was significantly longer in the ibrutinib cohort (median = not reached) than the ofatumumab cohort (median = 8.11 months; hazard ratio = 0.133; 95% CI 0.099-0.178). The three-year PFS rate was 59% and 3%, respectively.
The median treatment duration was 41 months in the ibrutinib cohort and 5 months in the ofatumumab cohort. The most common reasons for discontinuation of ibrutinib were progressive disease (27%) and AEs (12%). At median follow-up, 46% of patients in the ibrutinib cohort were still on therapy.
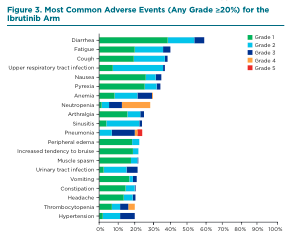
The most common grade 3 or higher hematologic AEs with ibrutinib were neutropenia (23%), anemia (9%), and thrombocytopenia (8%), and the most common grade 3 or higher nonhematologic AEs were pneumonia (17%), hypertension (8%), urinary-tract infection (6%), and diarrhea (6%). (See Figure 3.) No new safety signals have emerged in the study’s four-year follow-up. See Table 1 (bottom of page) for nursing best practices in managing AEs associated with ibrutinib.
Oncology nurses may be the first healthcare provider to observe treatment-related AEs and are instrumental in managing and educating patients about their course of treatment and expectations. Nurses can provide patients with tools to promote treatment adherence, such as pill diaries, online resources, and smartphone apps. The oncology nurse’s management approach should depend on the type and severity of an AE, how bothersome it is to the patient, and the risk-benefit profile. See sidebar for nursing strategies to monitor certain AEs in patients receiving ibrutinib.

Conversations With Experts
Q: What role do nurses play in managing AEs?
A: Nurses are the cornerstone for clinical care of adults with cancer, and specifically those with CLL/SLL. The nurse’s role is patient education from the time of diagnosis and throughout care when a patient is taking ibrutinib. At each patient encounter, it’s important to educate the patient and caregiver based on the symptom profile or AEs. Nurses need to help patients anticipate potential AEs so they are not alarmed if they experience diarrhea, fatigue, or nausea.
We know that CLL/SLL is a disease of older adults, so it’s important to ensure that they can retain the information. The nurse must determine how the patient best learns and deliver information accordingly.
Q: After reading the results of the long-term RESONATE trial, what were your thoughts?
A: I took away the major value and importance of collaborative teamwork between oncologists, nurses, and other healthcare providers to ensure that the patients’ symptoms are managed appropriately to avoid dose reductions or discontinuations.
Q: What are your best practices for managing the most common AEs observed in patients receiving ibrutinib?
A: One of the most common symptoms is gastrointestinal (GI) issues, such as diarrhea. For this AE, I would advise that patients take the medication at night to decrease GI upset. If they have diarrhea, I encourage them to drink more fluids and avoid anything that dehydrates. We all suggest that patients take antidiarrheal medication, as well.
Another AE is cytopenias: Neutropenia, thrombocytopenia, and anemia are not uncommon, and blood counts are monitored monthly.
Q: When a patient begins treatment with ibrutinib, what topics do you discuss to help set expectations?
A: It’s important to educate patients and reassure them this is a chronic disease. We help patients achieve long-term care goals and plans, help initiate them on medication, and speak to their family and caregivers about the diagnosis and management.
It’s important to set expectations: What does the symptom profile look like? What can they expect in the disease journey? How are they prepared to live with this chronic illness? Having that initial discussion on thoughts and emotions related to this diagnosis is important.
Q: Do you have any specific resources you point patients toward?
A: We have support groups for caregivers and patients. We have lay health advisors and a range of supportive resources with which to connect patients. We use the distress thermometer to determine their distress level and how we can appropriately address their needs. If the distress level is high, we need to figure out why: Is it financial? Are there employment concerns? These are key pieces in which nurses have a major role in providing a holistic approach to care, beyond just side effect management. We really want patients to have good quality years.
Q: What are your recommendations for patients who are receiving ibrutinib?
A: When patients come to the clinic, the first person they meet is usually the oncology nurse who will take their medical history, conduct a physical exam, and review the medication list. Once a diagnosis of CLL/SLL is confirmed, ibrutinib may be prescribed. It can be used for treatment in both frontline and relapsed CLL/SLL. If the drug is prescribed, the oncology nurse can help educate patients about the use of the medication, as well as expected AEs. AEs can be identified in an informed consent, which lists the majority of treatment-related AEs, as well as patient education materials. Common, minor AEs include myalgias, diarrhea, and infections.
Often, those more minor AEs go away with time. The longer a patient takes ibrutinib, the more likely the AEs will subside or lessen, and nurses should reassure patients that AEs are not permanent. Oncology nurses should stay current on ibrutinib-associated AEs and monitor patients for blood counts and severity of AEs. The trend of blood counts, as well as the severity of the AEs, may determine whether a dose adjustment is required.
Frequently, when patients get home or start taking a medication, they will have additional questions, so oncology nurses should provide a contact number for them to call. Oncology nurses can answer the questions over the phone or can consult their physician or advanced practice provider.
Q: What are your recommendations for other nurses when managing a patient receiving ibrutinib?
A: Patients should be educated on treatment-related AEs and encouraged to call their healthcare provider with any concerns. The severity of the AE will determine how quickly the patient needs to be seen. CLL/SLL is now a chronic illness, and treatment with ibrutinib enables patients to lead normal lives.
It is important that oncology nurses understand dosing considerations. For example, ibrutinib should be held for three days before and after a minor procedure and for seven days before and after a major procedure. It is important that patients feel comfortable discussing everyday occurrences with the oncology nurse to help manage ibrutinib AEs properly.
Q: Because ibrutinib is administered orally, how do you encourage treatment adherence?
A: The oncology nurse can discuss ibrutinib with patients and stress the importance of taking it every day. Each time we see patients, the oncology nurse should ask if they are taking ibrutinib daily, and, if not, try to determine the reason for nonadherence. Sometimes nonadherence can either be a misunderstanding about the medication, or it could be because of a major AE.
Q: Are there any resources, tools, or tips you provide to patients?
A: Even though AEs are listed in the consent form and patient education materials, I try to point out the more common AEs to patients when we meet in the clinic. Our facility has a patient information sheet on ibrutinib that discusses the medication and its associated AEs. I also direct them to the Pharmacyclics YOU&i program, which is a patient-access support program that can be accessed at imbruvicahcp.com/you-i-support-program.
Q: How do you keep an open dialogue with patients?
A: It is important to make patients feel comfortable at their clinic visits so they are more likely to contact you in the event of questions or concerns. In our clinic, we give business cards with our name, office telephone number, email address, and the MD Anderson address so patients have access to their healthcare team.
Q: What are your tips for discussing adherence to ibrutinib with patients, particularly if they are experiencing AEs while taking this drug?
A: When patients experienced an AE, I try to determine its severity and how it is affecting them. If it’s something like diarrhea, we can use supportive measures. If they are experiencing nausea with ibrutinib, I will recommend taking it at night so they can sleep through the AE, and I will prescribe antiemetics. I also evaluate the blood counts with the physician, and if they drop, a dose reduction of ibrutinib may be required to prevent the need for transfusion. And, for anything serious, such as bleeding, the oncology nurse can refer the patient to the closest emergency department.
Overall, an oncology nurse plays an important role in their patients’ lives and in the management of AEs from ibrutinib.







