Cortisol Biomarkers Help Researchers Understand Chemotherapy-Induced Peripheral Neuropathy
Clinicians and researchers know little about the mechanisms for chemotherapy-induced peripheral neuropathy (CIPN), a side effect of neurotoxic agents that can cause numbness, tingling, and pain in the upper and lower extremities. Until recently, mechanism-based treatment was difficult, but biomarkers are helping nurse scientists identify a potential connection to the hypothalamic-pituitary-adrenal (HPA) axis.
Using salivary measurements of cortisol levels, Knoerl et al (https://doi.org/10.1188/22.onf.207-211). conducted a study among 13 patients with breast, gastrointestinal, or gynecologic cancers and CIPN to begin to build the data for chronic stress and HPA axis dysregulation’s role in CIPN pathophysiology. They reported their findings (https://doi.org/10.1188/22.onf.207-211) in the May 2022 issue of the Oncology Nursing Forum.
Cortisol, HPA Axis, and CIPN
In response to stress, the body activates the HPA axis (https://doi.org/10.1188/22.onf.207-211) to release cortisol and lower inflammation. The HPA axis typically deactivates once cortisol binds to hippocampus receptors; however, during chronically stressful conditions such as ongoing neurotoxic chemotherapy treatment, it may begin producing dysfunctional cortisol (https://doi.org/10.1188/22.onf.207-211) that leaves peripheral inflammation unchecked and increases peripheral nociceptor sensitization.
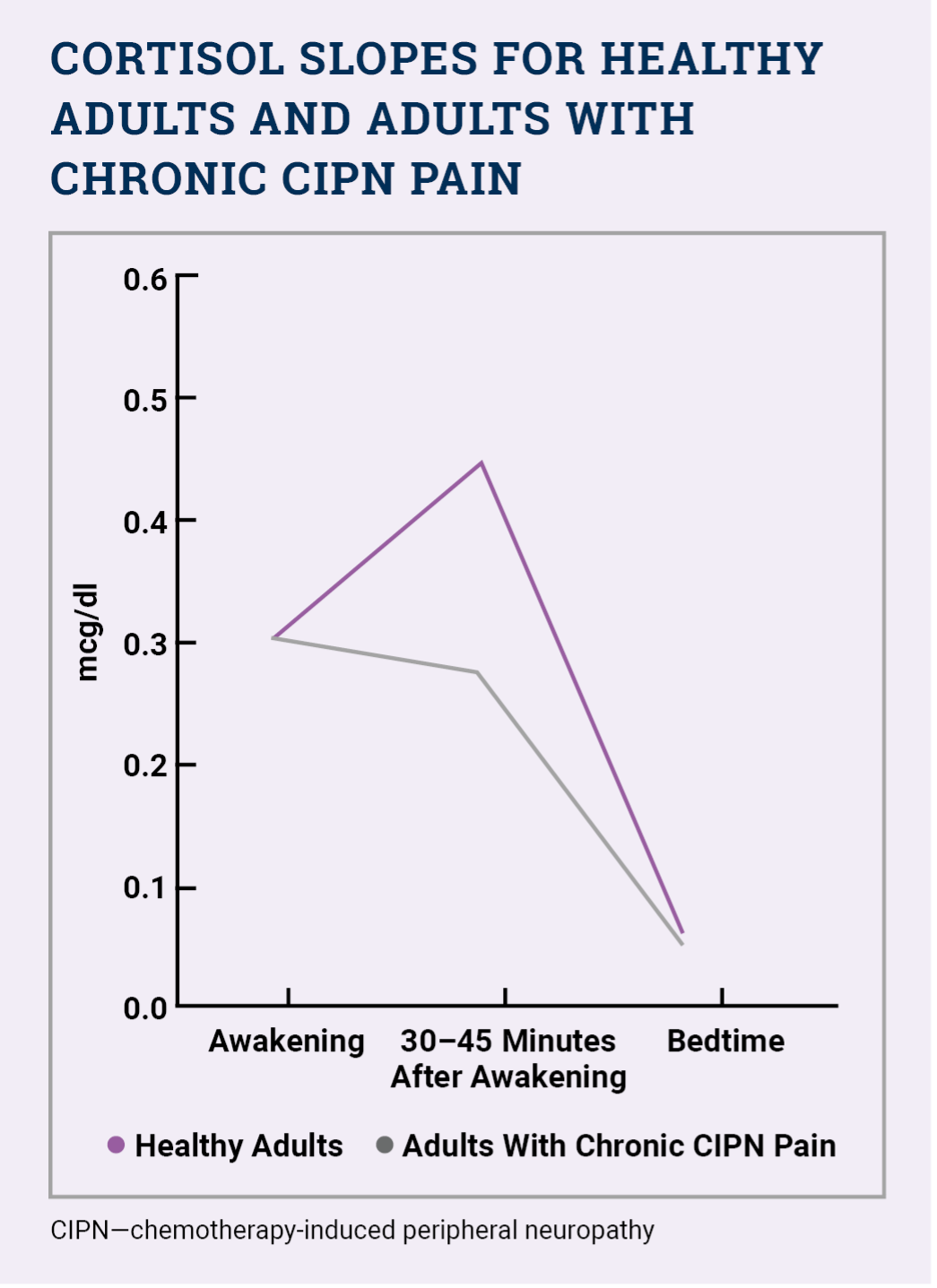
Salivary cortisol levels are therefore a useful biomarker to assess HPA axis functioning (https://doi.org/10.1188/22.onf.207-211). When the axis is healthy, a person’s morning cortisol levels rise by 50% approximately 30–45 minutes after awaking, but when it is dysfunctional, a person’s cortisol levels remain flatter.
Cortisol Levels in Patients Receiving Neurotoxic Chemotherapy
The researchers conducted a secondary analysis (https://doi.org/10.1188/22.onf.207-211) of data collected during their earlier pilot, randomized trial of a yoga intervention that obtained participants’ salivary cortisol samples. They analyzed cortisol for 13 patients with chronic CIPN (defined as persisting more than three months after chemotherapy) and a worst CIPN pain intensity score of 4 or higher during the past week on a numeric scale ranging from 0 (no pain) to 10 (worst pain).
Patients provided salivary samples when awaking, 30–45 minutes after awaking, and before bedtime across two consecutive days. The researchers calculated patients’ cortisol awakening response (i.e., change in cortisol from awaking to 30–45 minutes after awaking) and diurnal cortisol slope (i.e., difference in cortisol between awaking and bedtime). The median cortisol awakening response was –0.03 mcg/dl (range = –0.18 to 0.71), and the median diurnal cortisol slope was –0.24 mcg/dl (range = –0.42 to –0.05).
Overall, the researchers found that (https://doi.org/10.1188/22.onf.207-211) “cancer survivors with chronic CIPN pain exhibit an attenuated cortisol awakening response (about a 10% decrease) and flatter diurnal cortisol slope relative to healthy adults, potentially suggestive of HPA axis dysregulation” (see sidebar).
Research and Nursing Implications
The researchers collected the cortisol data from their parent study of a yoga intervention because the literature suggests that exercise may restore HPA axis regulation (https://doi.org/10.1188/22.onf.207-211) and reduce peripheral sensitization. Clinical studies have demonstrated that yoga significantly alters cortisol levels in individuals with chronic pain. However, their study occurred during the height of the COVID-19 pandemic and they were unable to obtain sufficient postintervention measurements to draw a conclusion about yoga’s effect on the HPA axis and cortisol.
Their data provides a baseline for future studies and demonstrates how biomarkers are used in cancer-related symptom management.
“The preliminary results of this study suggest that cancer survivors with chronic CIPN pain demonstrate an attenuated cortisol awakening response and flatter diurnal cortisol slope at baseline, suggesting that HPA axis dysregulation may be a contributing pathophysiologic mechanism to chronic CIPN pain,” the researchers concluded (https://doi.org/10.1188/22.onf.207-211). “Additional research in a larger sample of cancer survivors with chronic CIPN pain is needed to support the salivary cortisol patterns identified in this research. If further research supports the preliminary results, investigators may be able to measure the effectiveness of interventions targeting HPA axis dysregulation for the treatment of chronic CIPN pain.”
