Biologic agents or chemotherapies can trigger hypersensitivity reactions, which occur when a patient’s immune system identifies an agent as an antigen and initiates a complex reaction involving sensitization of T lymphocytes and macrophages, production of antibodies, and release of histamine. Developing and implementing a hypersensitivity kit containing all necessary medications and associated protocols allows nurses to immediately implement management when reactions occur.
The Evidence for Hypersensitivity Reaction Management Kits and Protocols
ONS member Marjorie Leslie, DNP, ACNP-BC, AOCNP®, director of clinical operations at Allegheny Health Network Cancer Institute in Pittsburgh, PA, said that her institution conducted a literature review to examine the evidence and best practices to ensure that their kits contained the necessary medications and devices. Leslie said that the literature describes:
- Best practices regarding anaphylactic reactions and management strategies
- Side effects of immunotherapy drugs and their management
- Hypersensitivity reactions to chemotherapeutic agents
- Diagnoses and management of drug hypersensitivity and anaphylaxis
- Management of hypersensitivity to specific cancer treatments and medications
Based on those findings, the team at Leslie’s institution developed and attached a hypersensitivity protocol order to every anticancer treatment plan, which allowed nurses to immediately respond to reactions. They built the kits in tackle boxes that stored necessary hypersensitivity management materials.
“Pennsylvania rules, regulations, and scope of practice require RNs to follow an order or an approved protocol for medication administration,” Leslie said. “Therefore, we had the written protocol approved by the necessary hospital committees to permit nurses to follow the protocol.”
They initially used a paper process, but implementation of their electronic health record system allowed them to apply hypersensitivity order sets to every treatment plan that were signed by the prescriber as PRN orders. Today, Leslie’s institution monitors every hypersensitivity reaction through a reporting database as an opportunity to track protocol compliance, patient outcomes, and trends.
Hypersensitivity Reaction Kits Reduce Delays in Management and Response
ONS member Valerie Quigley, BSN, RN, HN-BC, OCN®, ambulatory infusion manager at Valley-Mount Sinai Comprehensive Cancer Care in Paramus, NJ, said that oncology nurses at her institution are required to have their ONS/ONCC Chemotherapy Immunotherapy Certificate and trained to monitor and prepare for infusion reactions. However, the nurses identified a need for immediate standing orders for patients having or having the potential of a reaction.
Quigley explained that her institution’s infusion center has 23 infusion chairs and treats an average of 75 patients daily. An interprofessional team of nurses, pharmacists, and providers worked to create standing orders to allow oncology nurses to administer hypersensitivity medications based on the grade of the reaction.
“Hypersensitivity orders help to prevent delays in medication orders and responses,” Quigley said. “However, the nurses still had to go to our dispensing machine to remove the medication. The time for this step seems endless when a patient is having crushing back pain, shortness of breath, or other infusion-related reactions.”
Quigley said that her institution’s pharmacist recommended creating hypersensitivity reaction kits so oncology nurses had medications on hand immediately.
“At first, we only put the medications in our kits, which was only solving half of our problem,” Quigley said. “We recognized this quickly, and we were able to include medications and supplies that the nurse would need: alcohol wipes, syringes, EpiPens, methylprednisolone, diphenhydramine, and famotidine. The only medication that was not included was meperidine because it is a narcotic.”
The hypersensitivity reaction kits at Valley-Mount Sinai Comprehensive Cancer Care’s infusion center are clear, plastic boxes with tearaway locks similar to code cart locks. Quigley said that each kit is labeled with a clear expiration date and checked regularly by Valley pharmacy. Additionally, Quigley’s team created an adverse medication summary sheet to allow immediate documentation of vital signs, responses, and medications given, mimicking a code cart sheet to help nurses record the event.
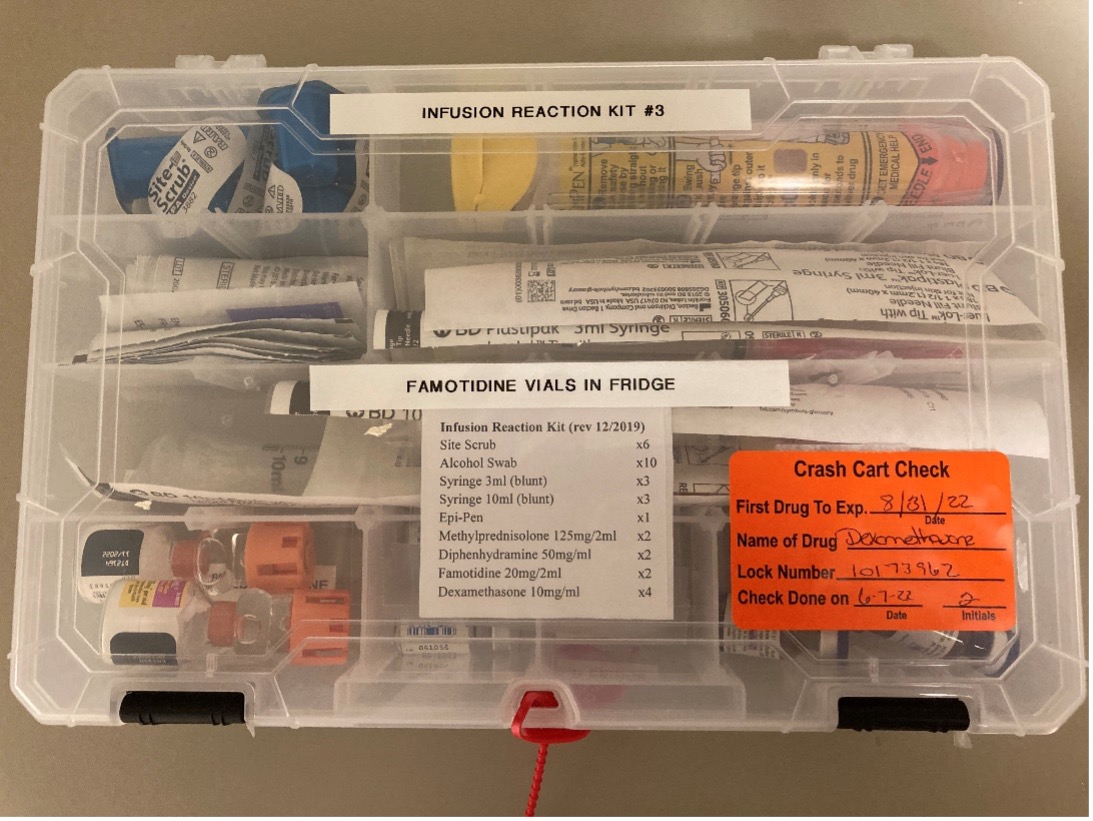
Quigley’s institution keeps two kits in each of its two medication rooms. Once a kit is opened, the nurse returns it to the pharmacy and a new kit is dispensed.
“The process of having hypersensitivity kits on hand and immediately available supports nurses’ autonomy and faster response times for our patients,” Quigley said. “Our charge nurse calls the covering provider to assess the patient and review the given medications.”
How You Can Implement Hypersensitivity Reaction Kits at Your Institution
Leslie emphasized the importance of training nurses on using hypersensitivity reaction kits when implementing a similar protocol, including direct guidance regarding the kit’s use, monitoring of protocol compliance, and periodic reeducation. She added that nurses and their institutions should stay up to date on current evidence to maintain the highest standard of practice.
Quigley advised beginning by developing and reviewing hypersensitivity orders that outline what medications nurses should give to patients according to each reaction grade. Then, build on those orders by asking your nurse colleagues which medications they need on hand or what they carry in their pockets. From there, you can customize your kit to your institution’s needs.
ONS members can join in on the conversation through ONS Communities.
Editor’s note: Leslie thanks ONS member Anna Vioral Gavin, PhD, Med, RN, OCN®, BMTCN®, director of oncology practice and professional development at Allegheny Health Network Cancer Institute in Pittsburgh, PA, for her assistance with the article.






